
Surmodics currently carries a Zacks Rank #3 (Hold). Per Medtronics website, IN.PACT Admiral DCB is the market-leading DCB that has demonstrated durable, consistent, and safe outcomes across multiple clinical trials. A 24-month data from the TRANSCEND study demonstrated continuous improvement in functional outcomes for treated patients, which was comparable to Medtronic’s IN.PACT Admiral DCB. SurVeil DCB also delivered a substantially lower drug dose versus MDT’s IN.PACT Admiral DCB. Data from the TRANSCEND 12-month pivotal clinical trial showed that SurVeil DCB met both the primary safety and primary efficacy endpoints that were found to be non-inferior compared to Medtronic’s IN.PACT Admiral DCB. 
TRANSCEND Study Dataĭata from the TRANSCEND study demonstrated compelling performance for SurVeil DCB. The study compared the results with Medtronic’s MDT IN.PACT Admiral DCB, which was evaluated in the comparator-arm, for non-inferiority. 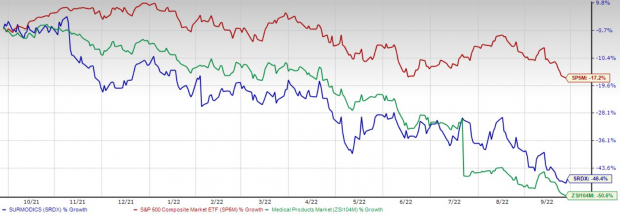
The study evaluated the safety and efficacy of SurVeil DCB, a next-generation device for the treatment of peripheral artery disease (PAD). The company completed the submission of the PMA application for SurVeil DCB in the third quarter of 2021, based on data from clinical trial - TRANSCEND. Surmodics announced last week that it has received a letter from the FDA indicating that a premarket approval (“PMA”) application for its SurVeil DCB is not currently approvable. The S&P 500 Index has declined 0.4% in the same time period. In fact, the company’s shares have declined 33.3% in the past six months against the industry’s 0.9% increase. SRDX have declined 31.8% since the close on Jan 18, following disappointing regulatory update related to its SurVeil drug-coated balloon (“DCB”).
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
